The most intuitive clinical trial software for growing teams
Clinical, Quality, and Regulatory content management systems built for scaling life science teams, who don’t have time for findings.





























Designed by people who know GxP compliance firsthand
Clinical trial compliance software and services for every team
Life science content management tech that helps small sponsors and CROs win big.

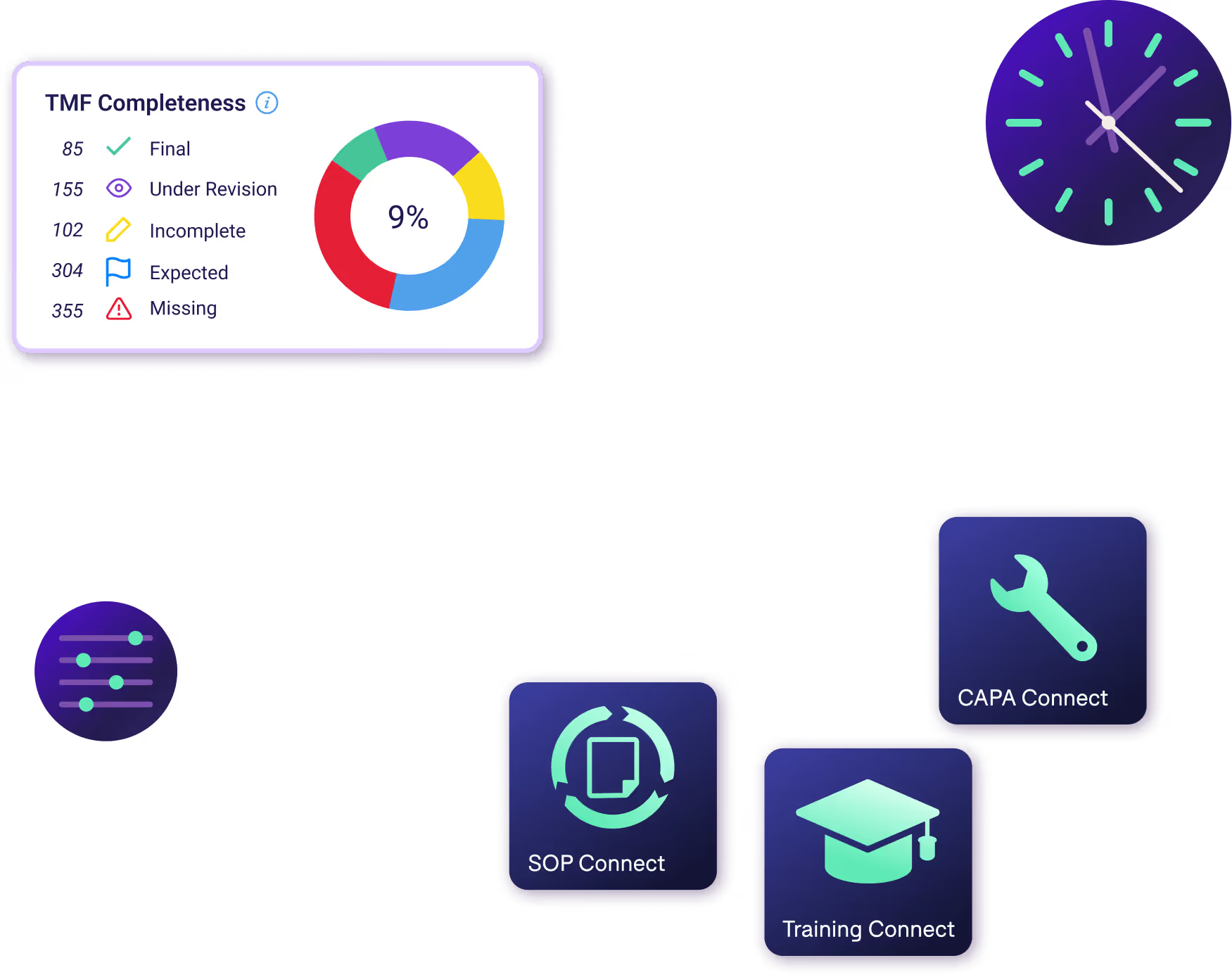
Tell a clear clinical trial story that inspectors can follow
Centralize every study record into one compliant hub, giving inspectors a complete, coherent view of your study — and your team the time back to focus on what actually matters to move the study forward.


Manage your entire quality system in one place
Bring document control, training oversight, and quality event management into one place, with the automated workflows to make sure nothing slips through the cracks.


Stop coordinating, start submitting
Centralize your entire regulatory operation, from submission planning to health authority correspondence, so lean teams move faster and never lose sight of what's holding things up.

Get — and stay — 21 CFR part 11 compliant
Ensure continuous validation of your e-signatures with every major Adobe Acrobat Sign Update. Montrium’s validation services perform the leg work, while you get the compliance stamp of approval from inspectors.

Technology gets you started. Support keeps you going.
Built for the teams running today’s clinical trials
From non-profits to BioPharma, Montrium gives scaling life sciences teams the confidence to know that their clinical, quality, and regulatory documentation are exactly where they need to be at the moment everything is on the line.

Nonprofit

Medical device

Pharma / Biotech

CRO
Speak with a product specialist
Tell us where your team is today and we'll show you what Montrium looks like for your specific situation.

Frequently asked questions
Find answers about Montrium product and services, implementation, and how it works for your organization.











